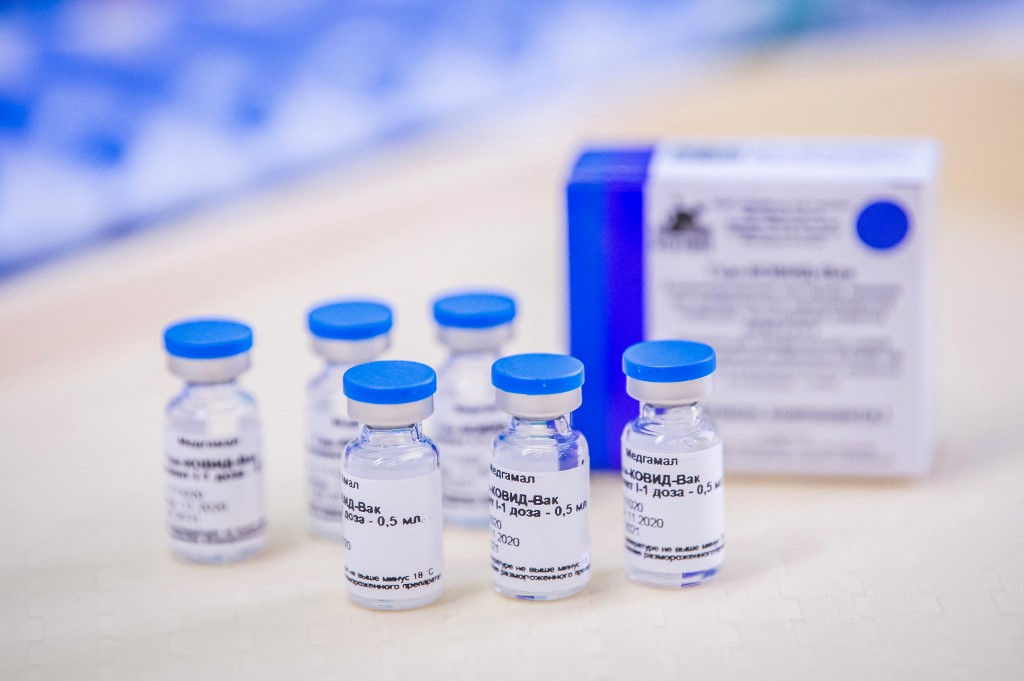
Russia hopes that Croatia will not delay procuring the Sputnik V COVID-19 vaccine, it was said on Friday on the Facebook profile of the Russian Embassy to Croatia.
Oglas
The Russian Embassy noted that intensive contact had been established between the vaccine manufacturer and Croatia's competent authorities, primarily the Health Ministry, expressing hope that there will be no delay in moving to practical implementation of the agreements reached.
Otherwise, "Croatian advisory services will be forced... to 'stand in line', waiting for consent from the EMA and the European Commission for as long as they determine", it was said in the statement.
The Croatian Agency for Medicinal Products and Medical Devices (HALMED) earlier said that it had received from the Ministry of Health documentation on the Sputnik V vaccine in order to ensure readiness for emergency import in case the need arose.
The European Medicines Agency (EMA) is conducting a gradual evaluation of the Sputnik V vaccine, in which HALMED experts are actively participating. The Lancet medical journal has recently published the results of clinical trials according to which the Russian vaccine is a safe and effective product in the fight against coronavirus.
Hungary was the first in the EU to approve Sputnik V and start administering the vaccine to its population. The same vaccine has sparked political turmoil in Slovakia and the Czech Republic, and Croatia, Latvia and Romania are currently waiting for EMA's green light to use the Russian vaccine.
"We have always said that health and life are above all and there can be no politics in in the supply of medicines... so it is important to join efforts to win the fight against the epidemic quickly," the Russian Embassy said.
Kakvo je tvoje mišljenje o ovome?
Pridruži se raspravi ili pročitaj komentare
Oglas
Kakvo je tvoje mišljenje o ovome?
Pridruži se raspravi ili pročitaj komentare
Oglas
NAJČITANIJE
Oglas
Oglas
Najnovije
Oglas



 Srbija
Srbija
 Bosna i Hercegovina
Bosna i Hercegovina
 Slovenija
Slovenija